
Aleks Engel
Partner, Novo Holdings and Director, REPAIR Impact Fund
Over the last 10 years, investors and big pharma have abandoned the field of antibiotic development because newly launched products generated poor revenues compared to other fields, such as oncology. Now is the time to re-enter.
The first principle of biopharmaceutical investors is to identify a match between innovation and unmet need. However, with about 200 approved antibiotics in 20 different drug classes, many needs were addressed for a while. Antimicrobial resistance (AMR) has slowly rendered older product classes obsolete, but with a combination of products that had become generic and thus cheap, infectious disease doctors had many available treatments at hand.
Today, new products are rarely used (also due to good stewardship to minimise new resistance development) and small companies manufacturing new products, such as Achaogen and Melinta, have gone bankrupt.
The onslaught of AMR has, however, continued and we now have close to a million deaths from drug-resistant infections per year, a number which keeps rising. Increasingly, older antibiotics have become inadequate, even in combinations.
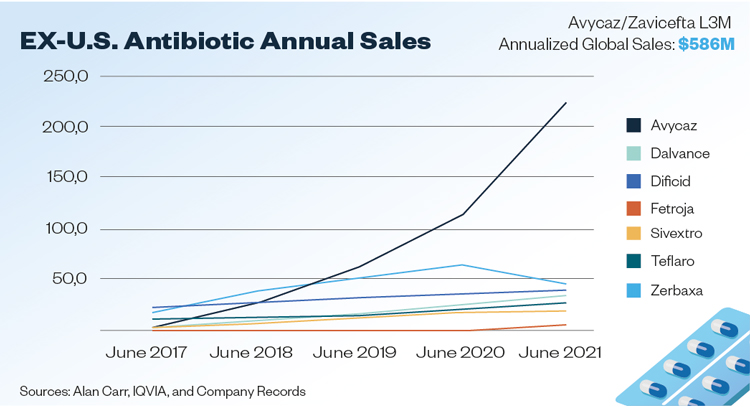
One new product, ceftazidime/avibactam, designed to circumvent certain bacterial resistance defences is selling greater than USD $500 million on a yearly run-rate1 and is on track to becoming the next antibiotic blockbuster. This is the data point that sceptical investors have been waiting for – proof that it is possible to make money with a differentiated product.
There are plenty of investment opportunities and the world is in dire need of these innovations becoming actual drugs that will save lives.
Another way to circumvent resistance is with the use of vaccines, avoiding the infection altogether. Vaccines (also against bacterial infections) are beginning to attract investors again. A good example was the EUR 47 million (USD 57 million) Series B investment in Minervax developing a vaccine against Strep B.
A good time to invest in innovation
The good news for investors considering stepping back into anti-infectives is that innovation is high. A recent pipeline review published in Nature2 found more than 400 early stage (pre-clinical) programs targeting high priority resistant pathogens.
There are plenty of investment opportunities and the world is in dire need of these innovations becoming actual drugs that will save lives.
We would love to hear from interested investors on [email protected]
[1] Based on L3M extrapolated from latest data ending June of 2021. Source: IQVIA
[2] https://www.nature.com/articles/s41579-019-0288-0


