Dr Jutta Amersdorffer
Medical Director (Consultant), Debiopharm International

Isabelle Petit
Global Value and Access Senior Manager, Debiopharm
An independent biopharmaceutical company is committed to developing tomorrow’s standard of care, with first-in-class novel antibiotic afabicin (Debio 1450) showing remarkable promise in bone and joint infection.
With surging AMR-infection coupled with a lack of alternatives to combat resistant pathogens,AMR is rapidly becoming a global public health emergency, a mission central to one Swiss biopharmaceutical company. Debiopharm’s Global Value and Access Senior Manager, Isabelle Petit, and Medical Director, Dr Jutta Amersdorffer, share how the company is helping in the fight against AMR.
The burden of antimicrobial resistance
“AMR is responsible for around one million direct deaths each year and associated with over four million more,”1 explains Petit. “Beyond this human toll, the economic impact could reach almost one trillion US dollars by 20501 if we fail to act.”
In addition, the difficulties in coding AMR as the main cause of death for patients who have multiple co-morbidities or the inconsistencies in coding method lead to an underestimation of the AMR-related mortality and hospitalisation period and mask the real-world burden of AMR.2
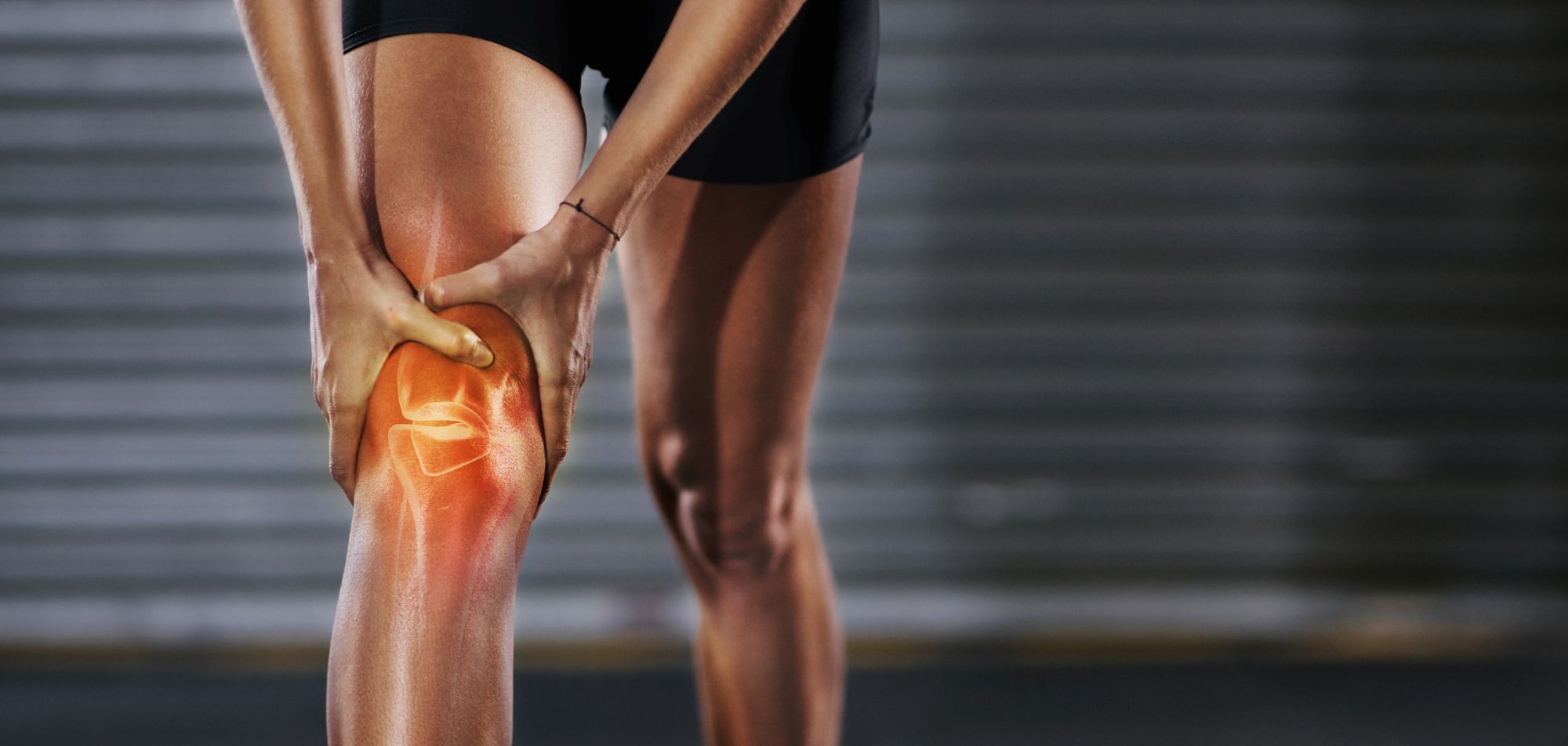
There have been no newly approved antibiotics for bone and joint infection in nearly 40 years,
Unmet need in bone and joint infections
One area of concern surrounds bone and joint infections, specifically those caused by bacteria, which have high morbidity and mortality rates.
“There have been no newly approved antibiotics for bone and joint infection in nearly 40 years,” says Dr Amersdorffer. “Patients often endure prolonged hospitalisation, and those with chronic infections may face amputation.”
The company is on a mission to change that, currently investigating the use of afabicin in a Phase 2 clinical trial for bone and joint infections.
Afabicin against AMR
Fulfilling all four WHO innovativeness criteria: new chemical class, new target, new mode of action and no cross-resistance to other antibiotic classes, afabicin (Debio 1450) is a first-in-class novel antibiotic that inhibits fatty acid synthesis in Staphylococci by targeting the Fabl enzyme.
“Afabicin has been designed to be highly specific,” explains Dr Amersdorffer. “It directly targets the source of infection, the Staphylococcus bacteria, without harming intestinal microbiota.” Afabicin’s narrow spectrum profile helps preserve the body’s healthy microbiome and reduce risk of further resistance, with early clinical data showing efficacy comparable to standard-of-care treatments.
“For Debiopharm, addressing antimicrobial resistance has always been a priority,” says Petit. “We recognised the urgent need for new antibiotics long before it became a mainstream concern, and that commitment has driven us to actively engage in this field and build strong in-house expertise.”
With the antibiotic market facing economic challenges, as the cost of development far outweighs its commercial return, it is key for companies like Debiopharm to collaborate with policy makers and industry to build consensus, define incentives to support antibiotic R&D and ensure that essential innovations like afabicin can be sustainably supported.
[1] WHO. (2023). Antimicrobial resistance. World Health Organization. https://www.who.int/news-room/fact-sheets/detail/antimicrobial-resistance
[2] Mestrovic T, Robles Aguilar G, Swetschinski LR, et al. The burden of bacterial antimicrobial resistance in the WHO European region in 2019: a cross-country systematic analysis. The Lancet Public Health. 2022;7(11):e897-e913. doi:10.1016/S2468-2667(22)00225-0
Invest, innovate and overcome drug resistance: https://www.debiopharm.com.